Sacrificial cathodic protection occurs when a metal is coupled to a more reactive (anodic) metal. This connection is referred to as a galvanic couple. In order to effectively transfer corrosion from the metal structure, the anode material must have a large enough natural voltage difference to produce an electrical current flow.
Effective application of cathodic protection can provide complete protection to any exposed areas for the life of the structure. The combination of an external coating and cathodic protection provides the most economical and effective choice for protection of underground and submerged structures. For bare or ineffectively coated existing structures,
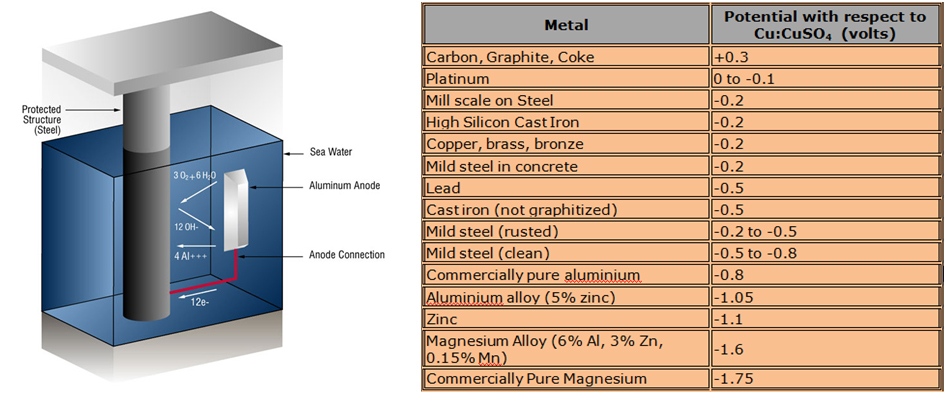
In order for galvanic cathodic protection to work, the anode must possess a lower (that is, more negative) electrode potential than that of the cathode (the target structure to be protected). The table below shows a simplified galvanic series which is used to select the anode metal. The anode must be chosen from a material that is lower on the list than the material to be protected.
Zinc and Aluminium are generally used in salt water, where the resistivity is generally lower. Typical uses are for the hulls of ships and boats, offshore pipelines and production platforms, in salt-water-cooled marine engines, on small boat propellers and rudders, and for the internal surface of storage tanks.
The advantages of sacrificial anode systems over others are they need no external power source, are easy to install, the low voltage and current between the anode and the surface it is protecting infrequently generates stray current, overprotection is unlikely, and inspection and monitoring is simple for trained personnel.
There are a few disadvantages including a limited current capacity based on the mass of the anode, ineffectiveness in high-resistivity environments. Increased weight on the protected structure, and increased air and water flow on moving structures such as ships.


